Home
| About
| Mine Tracker
| RSS
| Footer
▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄▄ █▄▄░▄▄█░▄▄▀█░▄▄▀█▀▄▀█░▄▄███░▄▄▀█░▄▄▀█░▄▀███▄▄░▄▄█░▄▄▀█░▄▄▀█▀▄▀█░▄▄█░▄▄▀█░▄▄▀██▄██░███▄██▄░▄█░██░ ███░███░▀▀▄█░▀▀░█░█▀█░▄▄███░▀▀░█░██░█░█░█████░███░▀▀▄█░▀▀░█░█▀█░▄▄█░▀▀░█░▄▄▀██░▄█░███░▄██░██░▀▀░ ███░███▄█▄▄█▄██▄██▄██▄▄▄███▄██▄█▄██▄█▄▄██████░███▄█▄▄█▄██▄██▄██▄▄▄█▄██▄█▄▄▄▄█▄▄▄█▄▄█▄▄▄██▄██▀▀▀▄ ▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀▀
FSMA Final Rule on Requirements for Additional Traceability Records for Certain Foods
https://www.fda.gov/food/food-safety-modernization-act-fsma/fsma-final-rule-requirements-additional-traceability-records-certain-foods
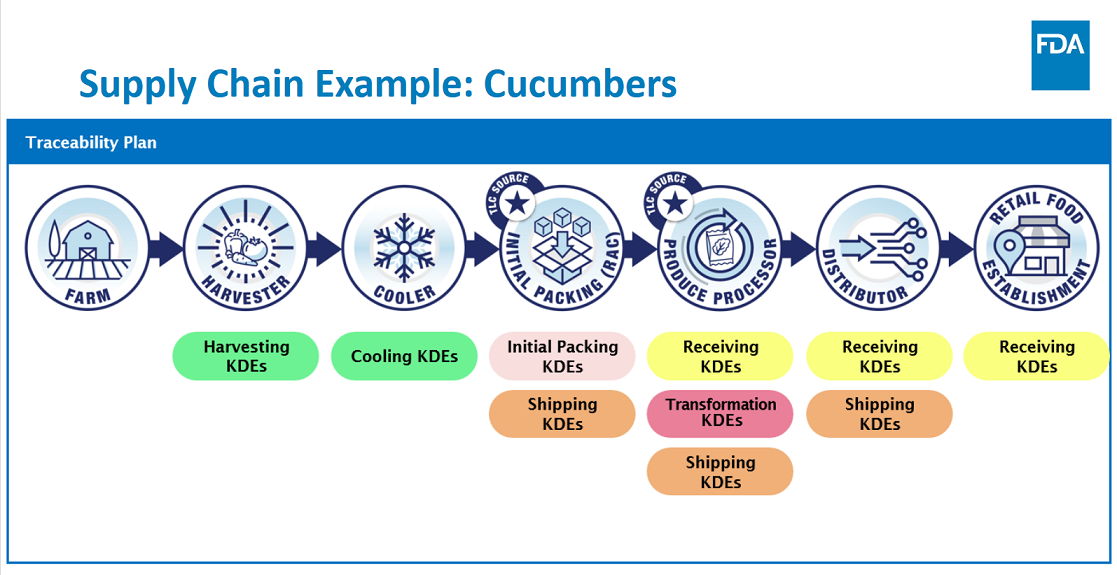
At the core of this rule is a requirement that persons subject to the rule who manufacture, process, pack, or hold foods on the FTL, maintain records containing Key Data Elements (KDEs) associated with specific Critical Tracking Events (CTEs); and provide information to the FDA within 24 hours or within some reasonable time to which the FDA has agreed.
https://www.fda.gov/food/food-safety-modernization-act-fsma/fsma-final-rule-requirements-additional-traceability-records-certain-foods

At the core of this rule is a requirement that persons subject to the rule who manufacture, process, pack, or hold foods on the FTL, maintain records containing Key Data Elements (KDEs) associated with specific Critical Tracking Events (CTEs); and provide information to the FDA within 24 hours or within some reasonable time to which the FDA has agreed.
WWF, Key Data Elements (KDE) and Traceability
https://wwflac.awsassets.panda.org/downloads/triptico_kde_en.pdf

2 page infographic / PDF
https://wwflac.awsassets.panda.org/downloads/triptico_kde_en.pdf

2 page infographic / PDF
Report, Key Data Elements for Seafood: A Compilation of Resources
https://fishwise.org/wp-content/uploads/2018/03/2017.05.25_KDEs-for-Seafood-Compilation-of-Resources_Final_-1-1.pdf
1 The purpose of this document is to compile existing publicly available key data element (KDE) resources regarding production, product identification, and supply chain traceability for seafood into one central location. This document does not address the full scope of KDEs that can be collected for seafood products (e.g. KDEs regarding food safety are not included) nor does it seek to define or clarify KDE definitions or terms. This document is intended for informational purposes only and should not be construed as legal advice nor as providing recommendations of any kind. Readers should always refer to the original reference source for complete information and important contextual background such as the scope and objectives of the specific resource. FishWise cannot be held liable for the accuracy or completeness of this document.
https://fishwise.org/wp-content/uploads/2018/03/2017.05.25_KDEs-for-Seafood-Compilation-of-Resources_Final_-1-1.pdf
1 The purpose of this document is to compile existing publicly available key data element (KDE) resources regarding production, product identification, and supply chain traceability for seafood into one central location. This document does not address the full scope of KDEs that can be collected for seafood products (e.g. KDEs regarding food safety are not included) nor does it seek to define or clarify KDE definitions or terms. This document is intended for informational purposes only and should not be construed as legal advice nor as providing recommendations of any kind. Readers should always refer to the original reference source for complete information and important contextual background such as the scope and objectives of the specific resource. FishWise cannot be held liable for the accuracy or completeness of this document.